Monatomic compounds are composed of single atoms and there are no chemical bonds between these atoms. Diatomic compounds are composed of molecules containing two atoms. ... The main difference between monatomic and diatomic compounds is the number of atoms present in those compounds.
Curtis Strite
Curtis Strite
A molecule that is made of just one atom, is called diatomic molecule.
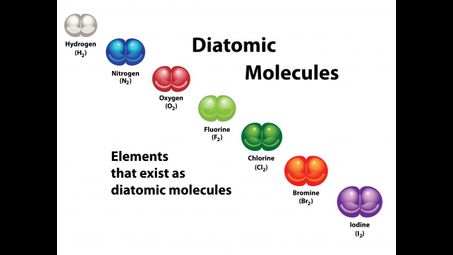
This is a list of the seven diatomic elements.
The seven diatomic elements are:
- Hydrogen (H2)
- Nitrogen (N2)
- Oxygen (O2)
- Fluorine (F2)
- Chlorine (Cl2)
- Iodine (I2)
- Bromine (Br2)
All of these elements are nonmetals, since the halogens are a special type of nonmetallic element. Bromine is a liquid at room temperature, while the other elements all gases under ordinary conditions. As the temperature is lowered or pressure is increased, the other elements become diatomic liquids.
Astatine (atomic number 85, symbol At) and tennessine (atomic number 117, symbol Ts) are also in the halogen group and may form diatomic molecules. However, some scientists predict tennessine may behave more like a noble gas.
While only these seven elements routinely form diatomic molecules, other elements can form them. However, diatomic molecules formed by other elements are not very stable, so their bonds are easily broken.
How to Remember the Diatomic Elements
The elements ending with "-gen" including halogens form diatomic molecules.
An easy-to-remember mnemonic for the diatomic elements is:
Have No Fear Of Ice Cold Beer
Michael Scalise
Monatomic
Add your answer:
What do you get when you combine1 hydrogen atom with one chlorine atom?
One molecule of a compound of hydrogen and chlorine that is called hydrogen chloride and has the formula HCl.
What is the smallest unit of salt that is still salt an atom or a molecule?
A molecule. One molecule of salt consists of one atom each of Sodium and Chlorine.
What does it mean if an atom has no subscript in a chemical formula?
There is one atom of that element in the molecule.
Is H2CO an atom element or molecule?
H2CO (I assume that the "2" is meant as a subscript of the "H") is a molecule called formaldehyde. It is called a molecule since it consists of more than one atombonded together (it consists of two hydrogen, one carbon, and one oxygen atom). Elements are made up of atoms; compounds are made up of molecules.
Is carbon dioxide molecule made of one carbon atom and one oxygen atom?
No. One carbon dioxide molecule is made up of one carbon atom and two oxygen atoms.
Is C an element or atom?
the answer is atom because even if it is the last one an atom is the smallest
How an atom relates to a molecule?
Molecules are formed by different arrangements of atoms. Example: one molecule of water is formed by 2 atoms of hydrogen and one atom of oxygen. (H2O). This is called the formula of a molecule.
What type of bonds forms a slightly positive hydrogen atom in one molecule and slightly negative atom in another molecule?
It is called hydrogen bond.
What type of bond form between a slightly positive hydrogen atom in one molecule and a slightly negative atom in another molecule?
This is called a hydrogen bond.
Heat is conducted when one atom or molecule collides with?
Heat is conducted when one atom or molecule collides with and transfers energy to another atom or molecule.
How is a molecule diffent from a atom?
A molecule contains more than one atom.
When elements have more than one atom what is it called?
Two atoms - Diatomic Molecule Three atoms - Triatomic Molecule
What do you get when you combine1 hydrogen atom with one chlorine atom?
One molecule of a compound of hydrogen and chlorine that is called hydrogen chloride and has the formula HCl.
Is neutralization reaction called displacements reaction?
No. A neutralization reaction is one in which an acid reacts with a base to form a salt and water. A displacement reaction is one in which one atom or molecule is replaced by another atom or molecule.
What does the element DO stand for?
There is no ELEMENT with the symbol "DO" however, it would be the molecule which is one atom of deuterium (D) and one atom of oxygen (O). Deuterium is an isotope of hydrogen. This molecule would be called deuterium oxide.
What is the smallest unit of salt that is still salt an atom or a molecule?
A molecule. One molecule of salt consists of one atom each of Sodium and Chlorine.
How can an atom not be a molecule?
A molecule is composed of one or more atoms that are properly bonded to each other. If a molecule contains more than one atom, each atom is then a partial molecule as opposed to a whole molecule. Therein lies the difference.
