I think this was based on the construction of Nitrogen.
Nitrogen has an atomic number of 7.
It has 2 electrons in it's first shell, leaving
5 electrons in it's outer shell.
It wants to fill the outer shell with 8 electrons so it can make 3 bonds with hydrogen.
So, one gets NH3. With each of the 3 electrons from Hydrogen bonded with a single electron from Nitrogen.
So, of the 5 electrons in Nitrogen's outer shell, 3 of them are bonded to Hydrogen, and 2 of them are just kind of sitting there.
This actually gives the Ammonia a tetrahedron shape. See the structure from Wikipedia:
http://en.wikipedia.org/wiki/Ammonia

With the two polar electrons up at the top.
Also, see the Wikipedia article on orbital hybridization.
http://en.wikipedia.org/wiki/Orbital_hybridisation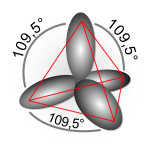
with four sp3 orbitals.
This extra sp3 orbital (pair of electrons) tends to attract a fourth Hydrogen (H+) ion.
Thus Ammonia (NH3) is polar, and acts as a base by attracting H+ ions, with its conjugate acid being Ammonium (NH4+)
What else can I help you with?
Why is ammonia polar covalent?
Ammonia is not a polar covalent molecule; it is a polar molecule. The nitrogen in ammonia is more electronegative than the hydrogen atoms, which leads to an uneven distribution of electrons in the molecule, creating a partial negative charge on the nitrogen and partial positive charges on the hydrogen atoms.
Is ammonia hydrophobic?
Ammonia is a polar molecule that can form hydrogen bonds with water molecules, so it is considered hydrophilic (water-loving) rather than hydrophobic (water-fearing).
Is ammonia a polar compound?
Yes, ammonia (NH3) is a polar compound. Although it has a pyramidal shape, the asymmetrical distribution of the nitrogen and hydrogen atoms results in an uneven charge distribution, creating a polar molecule with a net dipole moment.
Is ammonia soulible in hexane?
No, ammonia is not soluble in hexane. Ammonia is a polar molecule while hexane is nonpolar, so they do not mix well together.
Why is ammonia considered a polar molecule?
Ammonia is a polar molecule because the different electronegativity's of the nitrogen and the hydrogen molecules makes the hydrogen slightly positive and the nitrogen slightly negative. However there are 2 valance electons of the nitrogen atom which are not bonded to anything, which are called lone pairs. The lone pairs means that the ammonia molecule is not symmetrical therefore the electronegativity's do not cancel eachother, creating a polar molecule. ( The reason why the shape is no longer symmetircal is because lone pairs repel more than the bond pairs of the N-H, and essentially the bonding pairs are "squeezed" together, which accounts for the smaller than expected bond angle of 107 degrees)
What is an example of a polar molecule other than water?
Ammonia (NH3) is an example of a polar molecule. Its trigonal pyramidal shape results in an unequal distribution of charge, making it a polar molecule.
Is H plus polar or non polar?
If you think to ammonia molecule, this is polar.
Is ammonia mlecule NH3 more polar than water molecule?
No, water molecule (H2O) is more polar than ammonia (NH3) because of the greater difference in electronegativity between oxygen and hydrogen atoms in water molecule compared to nitrogen and hydrogen atoms in ammonia molecule. Water has two polar covalent bonds while ammonia has only one.
Why is ammonia polar covalent?
Ammonia is not a polar covalent molecule; it is a polar molecule. The nitrogen in ammonia is more electronegative than the hydrogen atoms, which leads to an uneven distribution of electrons in the molecule, creating a partial negative charge on the nitrogen and partial positive charges on the hydrogen atoms.
Is ammonia hydrophobic?
Ammonia is a polar molecule that can form hydrogen bonds with water molecules, so it is considered hydrophilic (water-loving) rather than hydrophobic (water-fearing).
Is ammonia is soluble in water or a polar molecule?
Ammonia form in water ammonium hydroxide - NH4OH.
Is ammonia soluble in propylene?
Ammonia is generally not soluble in propylene, which is a nonpolar hydrocarbon. Ammonia is a polar molecule, and its solubility is better in polar solvents like water. The lack of significant interaction between the polar ammonia and nonpolar propylene limits its solubility in the latter.
Is ammonia a polar compound?
Yes, ammonia (NH3) is a polar compound. Although it has a pyramidal shape, the asymmetrical distribution of the nitrogen and hydrogen atoms results in an uneven charge distribution, creating a polar molecule with a net dipole moment.
Is ammonia soulible in hexane?
No, ammonia is not soluble in hexane. Ammonia is a polar molecule while hexane is nonpolar, so they do not mix well together.
Why is ammonia considered a polar molecule?
Ammonia is a polar molecule because the different electronegativity's of the nitrogen and the hydrogen molecules makes the hydrogen slightly positive and the nitrogen slightly negative. However there are 2 valance electons of the nitrogen atom which are not bonded to anything, which are called lone pairs. The lone pairs means that the ammonia molecule is not symmetrical therefore the electronegativity's do not cancel eachother, creating a polar molecule. ( The reason why the shape is no longer symmetircal is because lone pairs repel more than the bond pairs of the N-H, and essentially the bonding pairs are "squeezed" together, which accounts for the smaller than expected bond angle of 107 degrees)
What is a molecule with opposite charge on each end called?
A molecule with opposite charge on each end is called a polar molecule. Examples include water (H2O) and ammonia (NH3).
Is NH3 polar or non-polar?
NH3, or ammonia, is a polar molecule due to its trigonal pyramidal geometry and the presence of a lone pair of electrons on the nitrogen atom. This lone pair induces a net dipole moment in the molecule, making it polar.
