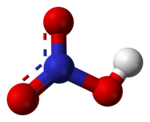
Look at the nitric acid on Wikipedia.
http://en.wikipedia.org/wiki/Nitric_acid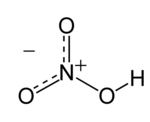


What else can I help you with?
____ HNO3 + ____ KOH → ?
HNO3(aq) + KOH(aq) --> KNO3(aq) + H2O(l) Because HNO3 is a strong acid, when it combines with a strong base it will react. When you deal with a problem like this the positive ions, H+ and K+ switch places.
What is the oxidation numbers of HNO3?
The oxidation numbers of each element in HNO3 are: H is +1, N is +5, and O is -2. The sum of the oxidation numbers in a neutral compound like HNO3 must equal zero.
What is KOH HNO3 balanced?
HNO3(aq) + KOH(aq) --> KNO3(aq) + H2O(l) Because HNO3 is a strong acid, when it combines with a strong base it will react. When you deal with a problem like this the positive ions, H+ and K+ switch places.
Nitric acid is represented by the chemical formula?
I believe it is HNO3
Chemical formula for nitric acid?
Nitric Acid is HNO3The chemical formula for nitric acid is HNO3
____ HNO3 + ____ KOH → ?
HNO3(aq) + KOH(aq) --> KNO3(aq) + H2O(l) Because HNO3 is a strong acid, when it combines with a strong base it will react. When you deal with a problem like this the positive ions, H+ and K+ switch places.
What is the oxidation numbers of HNO3?
The oxidation numbers of each element in HNO3 are: H is +1, N is +5, and O is -2. The sum of the oxidation numbers in a neutral compound like HNO3 must equal zero.
What is KOH HNO3 balanced?
HNO3(aq) + KOH(aq) --> KNO3(aq) + H2O(l) Because HNO3 is a strong acid, when it combines with a strong base it will react. When you deal with a problem like this the positive ions, H+ and K+ switch places.
What is the name of HNO3?
HNO3 is Nitric Acid
Nitric acid is represented by the chemical formula?
I believe it is HNO3
What is the acid for HNO3?
HNO3 is called nitric acid.
Is HNO3 neutralized?
HNO3 is not neutral.It is a strong acid.
What is the formula of hydrogen nitrate?
Hydrogen Nitrate ???? H-NO3 Is more usually known as Nitric Acid and has the formula 'HNO3'.
Chemical formula for nitric acid?
Nitric Acid is HNO3The chemical formula for nitric acid is HNO3
What is the molecular equation for HNO3 plus KOH?
The molecular equation for the reaction between nitric acid (HNO3) and potassium hydroxide (KOH) is: HNO3 + KOH -> KNO3 + H2O.
What is the scientific name for HNO3?
The scientific name for HNO3 is nitric acid.
What is the formula for nitric acid?
Nitric Acid ; HNO3 Do NOT confuse with Nitrous Acid ; HNO2 NB Note the different suffixes and the different number of oxygen atoms.
